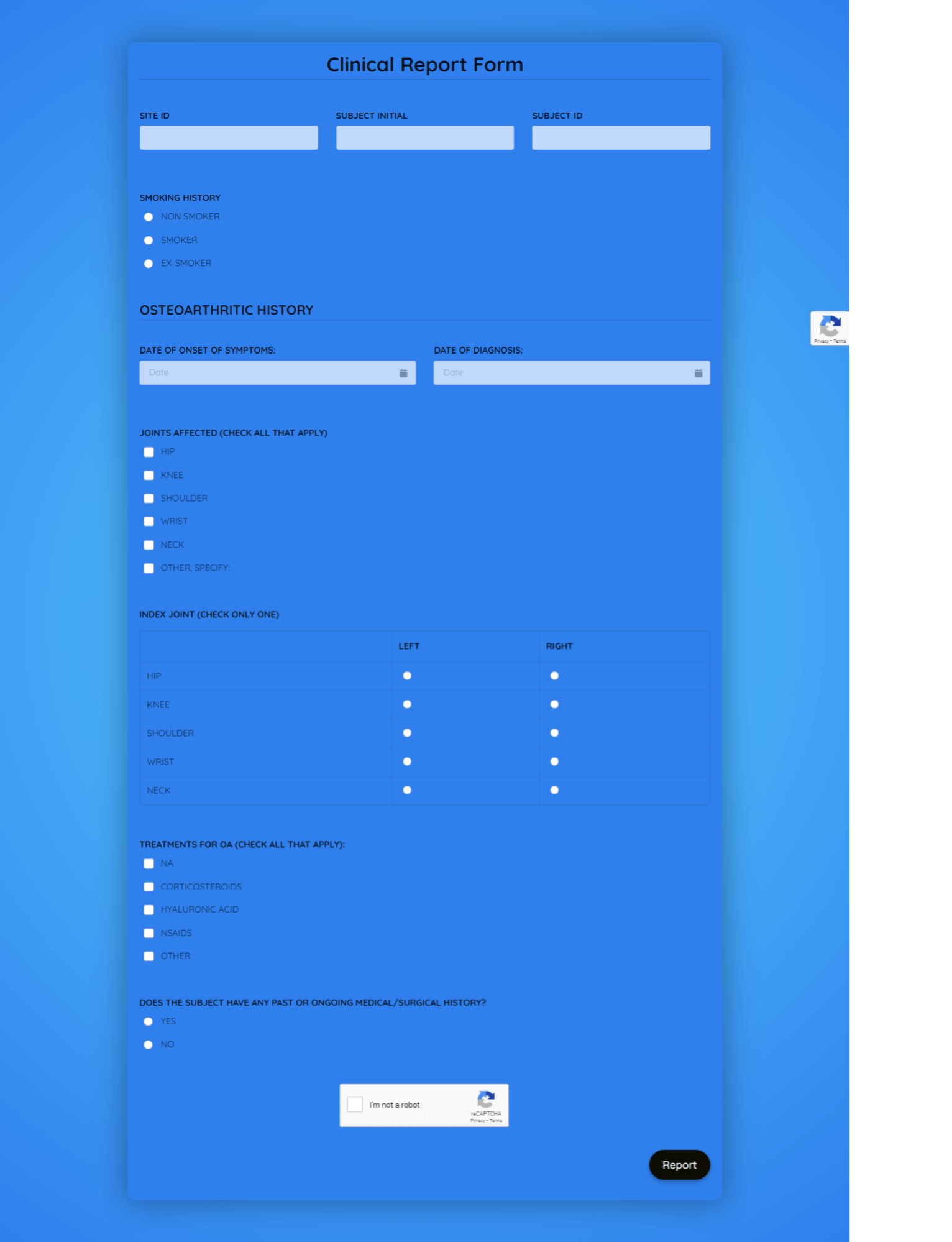
Clinical Report Form Template
A clinical report form is a pivotal part of the clinical research development. The data gathered from the clinical report form can be analyzed and used to draw conclusions to aid the clinical research. This generic clinical report form can be tailored to meet your goals. Get started with this form today.
Use this template Tips for using template
Type
Industry
- Agency Form
- Banking Form
- Business Form
- Customer Service Form
- Ecommerce Form
- Education Form
- Event Form
- Health Form
- Human Resources Form
- Legal Form
- Marketing Request Form
- Non Profits
- Photography Form
- Real Estate Form
- Research Form
- Sport Form
- Accounting Form
- 10 Free Advertising Form
- 84 Free Agreement Form
- 102 Free Application Form
- Appointment Form
- Free Appraisal Form
- 10 Free Approval Form
- 72 Assessment Form
- 10 Free Attendance Form
- 87 Free Booking Form
- Free Calculation Form
- Christmas Form
- Claim Forms
- Client Form
- 39 Complaint Form
- Conditional Logic
- 12 Online Consent Form
- 33 Free Construction Form
- 33 Consultation Form
- Contact Form
- 69 Free Contract Form
- 12 Customer Feedback
- Customised Form
- 55 Free Data Collection Form
- Employment Form
- Enquiry Form
- 70 Evaluation Form
- 21 Free Event Survey
- Exit Survey
- 45 Expense Form
- 96 Feedback Form
- File Upload Form
- Financial Form
- Halloween Forms
- Information Form
- 100 Free Inspection Form
- 63 Free Intake Form
- 55 Free Lead Generation Forms
- 33 Free Leasing Form
- 15 Free Maintenance Form
- 40 Market Research Survey
- 45 Free Medical Form
- 57 Online Membership Form
- Multi Page
- 33 Free Order Form
- 37 Payment Form
- 60 Survey Questionnaire
- Quizzes
- 21 Referral Form
- 105 Free Registration Form
- 35 Free Release Form
- 30 Rental Form
- Repeating Data
- 70 Free Report Form
- 153 Free Request Form
- 24 Free Requisition Form
- 35 Reservation Form
- 50 Free Rsvp Form
- 33 Free Satisfaction Survey
- 45 Free School Form
- 100 Free Sign Up Form
- 21 Sponsorship Form
- 75 Free Survey Form
- Free Therapy Form
- 70 Free Tracking Form
- Training Form
- Verification Form
- 25 Free Waiver Form
By Type
By Industry
- Agency Form
- Banking Form
- Business Form
- Customer Service Form
- Ecommerce Form
- Education Form
- Event Form
- Health Form
- Human Resources Form
- Legal Form
- Marketing Request Form
- Non Profits
- Photography Form
- Real Estate Form
- Research Form
- Sport Form
- Accounting Form
- 10 Free Advertising Form
- 84 Free Agreement Form
- 102 Free Application Form
- Appointment Form
- Free Appraisal Form
- 10 Free Approval Form
- 72 Assessment Form
- 10 Free Attendance Form
- 87 Free Booking Form
- Free Calculation Form
- Christmas Form
- Claim Forms
- Client Form
- 39 Complaint Form
- Conditional Logic
- 12 Online Consent Form
- 33 Free Construction Form
- 33 Consultation Form
- Contact Form
- 69 Free Contract Form
- 12 Customer Feedback
- Customised Form
- 55 Free Data Collection Form
- Employment Form
- Enquiry Form
- 70 Evaluation Form
- 21 Free Event Survey
- Exit Survey
- 45 Expense Form
- 96 Feedback Form
- File Upload Form
- Financial Form
- Halloween Forms
- Information Form
- 100 Free Inspection Form
- 63 Free Intake Form
- 55 Free Lead Generation Forms
- 33 Free Leasing Form
- 15 Free Maintenance Form
- 40 Market Research Survey
- 45 Free Medical Form
- 57 Online Membership Form
- Multi Page
- 33 Free Order Form
- 37 Payment Form
- 60 Survey Questionnaire
- Quizzes
- 21 Referral Form
- 105 Free Registration Form
- 35 Free Release Form
- 30 Rental Form
- Repeating Data
- 70 Free Report Form
- 153 Free Request Form
- 24 Free Requisition Form
- 35 Reservation Form
- 50 Free Rsvp Form
- 33 Free Satisfaction Survey
- 45 Free School Form
- 100 Free Sign Up Form
- 21 Sponsorship Form
- 75 Free Survey Form
- Free Therapy Form
- 70 Free Tracking Form
- Training Form
- Verification Form
- 25 Free Waiver Form
Clinical Report Form Template
What is a Clinical Report Form (CRF)
A Clinical Report Form (CRF) is a structured document used in clinical research and trials to collect data about participants' medical history, treatments, outcomes, and other relevant information. CRFs serve as a standardized tool for documenting and tracking data throughout a clinical study. They are crucial for maintaining accuracy, consistency, and compliance in clinical research.
Two Types of CRF:
- Paper CRF: This traditional type involves physical paper forms that are filled out manually by clinicians or research staff during patient visits. The completed forms are then usually entered into a digital database for analysis.
- Electronic CRF (eCRF): In this digital age, eCRFs are becoming more common. They are digital forms accessed and filled out electronically using specialized software or platforms. eCRFs streamline data collection, reduce errors, and enable real-time data entry and analysis.
Creating a Clinical Report Form Using Formplus Form Template:
Creating a clinical report form using Formplus is a straightforward process. Formplus is an online form builder that offers customizable templates. Here's a basic guide:
- Access Formplus: Go to the Formplus website (formpl.us) and sign up for an account.
- Choose Template: Browse through the available templates or use the search bar to find a "Clinical Report Form" template.
- Customize Form: Once you've selected a suitable template, customize it to match the specific data fields required for your clinical study. Common fields include patient information, medical history, treatment details, adverse events, and follow-up assessments.
- Data Validation: Implement data validation rules to ensure that the entered data follows the required format and standards.
- Integrate Features: Formplus offers features like conditional logic, which displays specific fields based on user responses. Utilize such features to create a dynamic and user-friendly form.
- Notifications and Data Storage: Configure notification settings to receive alerts when new data is submitted. You can also integrate the form with other tools for data storage and analysis.
- Publish: After customization, publish the form to get a shareable link or embed code for distribution to clinical staff or research participants.
Contents of a Clinical Study Report:
A comprehensive clinical study report typically includes:
- Title Page: Identifies the study, its authors, and the date of compilation.
- Table of Contents: Lists the sections and their respective page numbers for easy navigation.
- Executive Summary: Provides a concise overview of the study's objectives, methodology, key findings, and conclusions.
- Introduction: Explains the study's rationale, objectives, and context.
- Methods: Details the study design, participant selection, interventions, data collection, and statistical analysis plan.
- Results: Presents the collected data, including tables, graphs, and statistical analyses.
- Discussion: Interprets the results, compares them with existing research, and discusses implications.
- Conclusion: Summarizes the study's findings, significance, and potential future directions.
- References: Lists the sources cited throughout the report.